TOP > Report & Column > The Forefront of Space Science > 2015 > The Liquid Boron Turned Out to Be a Semiconductor
![]()

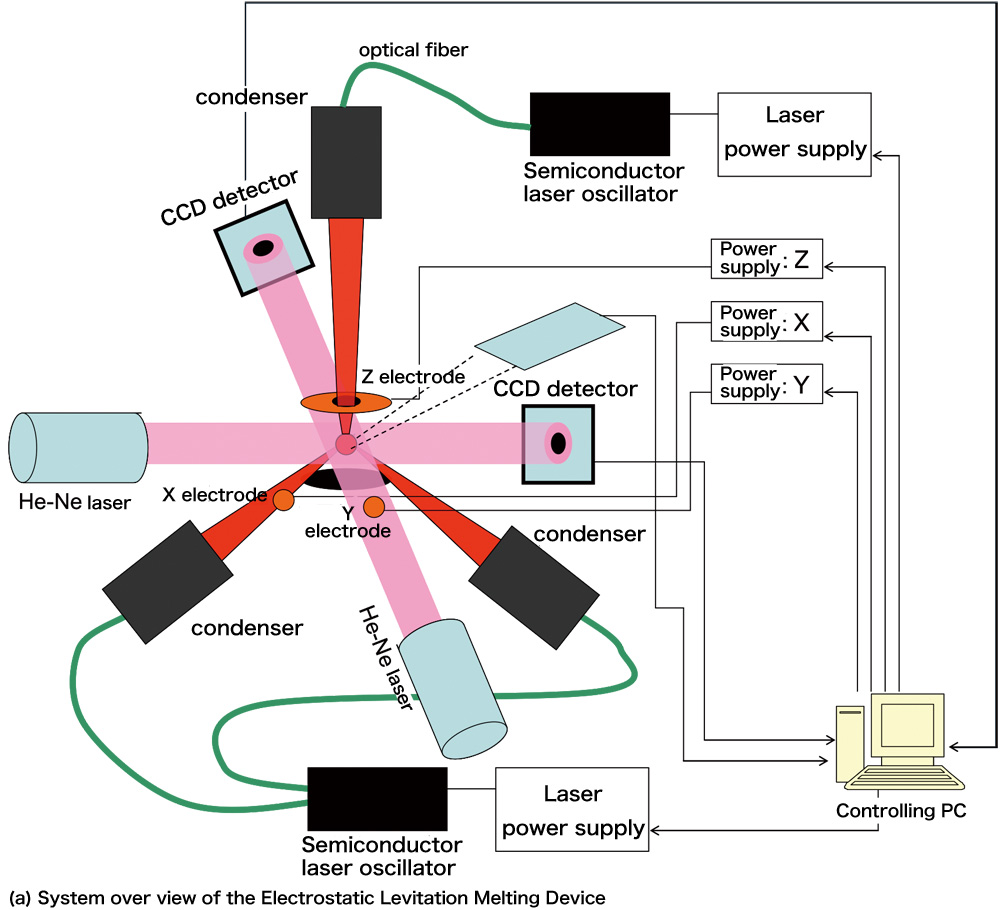
Electrostatic Levitation Technique To deal with liquids on the ground, we need a container to hold the liquids. However, while holding liquids with a container, impurities contamination from the container wall or the reactions between the liquid and the container will cause problems. The container-less processing (levitation), which has been put into practice recently, is drawing a great deal of attention as an epochal method without causing problems of containers. The Electrostatic Levitation Technique developed by us uses Coulomb's force to levitate the samples. As shown in figure 2 (a), when we apply an electric field to a charged sample and balance the force and the gravity, the sample will be able to levitate at any place between the two electrodes. The levitating sample will melt down when heated with laser. The size of the sample is about 2mm while the distance between the standard electrodes is about 10mm. A voltage of 10~20kV is applied between the electrodes and to prevent from discharging, a vacuum is kept inside the chamber. Two CCD position detectors are used to detect the three dimensional position of the sample. With the position information measured, we adjust the voltage between the electrodes with PID Controller (Proportional-Integral-Derivative Controller, a feedback controller) so that we can stabilize the position of the sample with an accuracy within ±10μm. We measure the temperature of the sample with a radiation thermometer. No matter whether the sample is a metal or an insulator, it can be levitated as long as it is charged. If we raise the output of laser, we will even be able to melt down the tungsten (W, melt point: 3422°C). As to the boron, we can melt it while keeping it in the air like figure 2 (b).
X-ray Compton Scattering Experiment Although we have been able to keep the boron melt with the use of Electrostatic Levitation Technique, there comes the next problem of how to investigate whether the melted boron is a metal. In general, to investigate whether a substance is a metal or not, we research how it conducts the electricity (conductimetry). To do that, we have to attach more than two electrodes to the substance and see the relationship between the current and voltage between the two electrodes. It is easy for us to attach the electrodes to solid substances and measure. Even for the liquids, we are as well able to measure with electrodes inserted in the liquid, if the melting point and the reactivity are both not high. However, since we have not found any substance that does not react with the boron melt, there is no electrode to insert in it and we are not able to measure the conductance of boron melt. Therefore, we decided to research the actions of the electrons in boron melt with X-ray. The X-ray, a form of electromagnetic radiation, will strongly interact with electrons. When an X-ray is irradiated to the substance, we can find the actions of the electrons in the substance by researching the details of the scattered X-ray. Since we have to use a very powerful x-ray during real measurements, we decided to use an experiment method called Compton scattering measurement with the large synchrotron radiation facility SPring-8. Figure 3 shows the experiment. We sent the whole Electrostatic Levitation Melting Device from the Tsukuba Space Center to the SPring-8 located in Sayou-chou, Hyogo Prefecture, assembled the device in the beamline and conducted the experiment.
|
||||||||||