TOP > Report & Column > The Forefront of Space Science > 2014 > Removing the Carbon Dioxide Using Fuel Cells
![]()

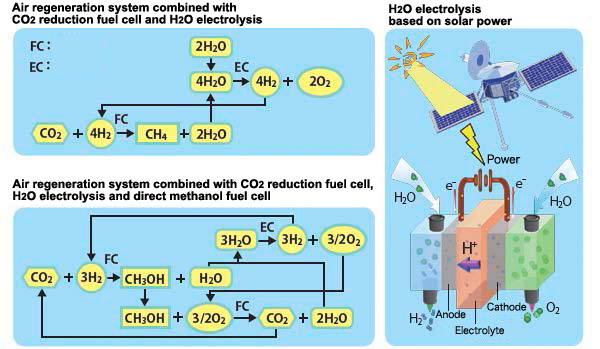
Next Generation Air Regeneration System As mentioned at the beginning, in the closed spaces with human activities, it becomes necessary to remove CO2 and currently we are recovering CO2 by absorption. However, from a long-term perspective, we should proactively recycle the CO2. Here we firstly talk about a next generation air regeneration system for closed spaces using the hydrogen-carbon dioxide fuel cell. The upper left of figure 3 shows the system combined with the CO2 reduction, based on equation (2), and the electrolysis of H2O (reverse reactions of the equations 3.1 and 3.2), which is one of the products of the reduction. Since the equation (2) has been introduced in figure 2, here we omit some details. One of the products, H2O, will be put into electrolysis. Since the electrolysis of water is an endothermic reaction and needs external energy, so we cater this with solar power (Figure 3 right). The O2 obtained from the electrolysis of H2O will be used for life activity (breathing), and the H2 will be reused in CO2 reduction. However, the H2 produced is not enough for the equation 2 to proceed, so we have to rely on the H2O replenished from outside for the insufficient H2. And the other product of CO2 reduction, CH4, will be emitted out of the system.
So far, we have been describing the case when the main product of CO2 reduction is CH4, but there are other byproducts among the reductants as well, and the types of those byproducts will differ with the electrode catalyst and working conditions of the fuel cells. So then, the lower left of figure 3 shows another system when there is supposed to be methanol (CH3OH) in the products. Though the CO2 reduction and the H2O electrolysis in the lower left of figure 3 are same as the upper left, the CH3OH produced can be used as fuel for other fuel cells. This is called direct methanol fuel cell, a power generation technology which has been established on the ground. So, we can get a new air regeneration system by combining these 3 technologies together. In addition, we can put the CH3OH into storage and use it as a spare power supply when the solar power is unavailable. Prospects of Space Applications Toward the practical application of this technology, we will need to solve several important issues. As mentioned above, to make general chemical reactions proceed rapidly, it is effective to use suitable catalysts. For reactions on the electrode (electrode reaction), it is not any conductive material can be used as electrodes. It is necessary to develop electrode catalysts which can minimize the energy loss and control the types of products selectively. On the other hand, when we consider the long-term space stay, we have to consider a larger carbon circulatory system in addition to the oxygen circulation. Focusing on the CO2, CH4, CH3OH, H2O, H2 and O2 involved in each reaction so far, it seems to be essential to take organic synthesis and food synthesis into consideration as well. In the ancient Earth, CO2, which accounted for most of the air, has been converted into O2 and organic matter by microorganisms. It must be worthwhile to increase the technological capability to artificially make it able to carry out this in other planets or satellites. We expect that the development of this study will be useful in space development in some form or other in the distant future.ÍÍ In Conclusion Since the Lehman shock, advanced research seems to be globally decreasing. Among the studies, the joint research on hydrogen-carbon dioxide fuel cells with ISAS is expected to be a dream technological development as a medium and long term theme from the near future. Last but not least, this research is still on going under the joint work with the ISAS. Part of this research is conducted by the Strategic Basic Research Programs (ACT-C) of the Japan Science and Technology Agency. I would like to thank immensely to the parties concerned. (Minoru Umeda)
|
||||||