TOP > Report & Column > The Forefront of Space Science > 2009 > Research on Ultra-High Temperature Liquids Using the Electrostatic Levitation Technique
![]()

If these events actually arise in liquid silicon, the picture of crystal growth in the solid-liquid interface would change greatly and become clearer. Despite a number of experiments to date, however, no one has succeeded in witnessing directly the presence of covalent bonds. This is because many difficulties remain to be cleared. For example, the melting point of silicon is very high (1,412 deg. C) so it is difficult to retain stably liquid silicon, which has high chemical responsiveness. We also had a problem of finding an appropriate measurement method to observe directly the electron status of high-temperature samples. These problems made experimental research difficult. By combining the electrostatic levitation technique and radiation, we were able to overcome these hurdles successfully. Using X-ray (116keV) radiation of the SPring-8, we measured the Compton scattering from clean liquid silicon levitated in a vacuum environment by the electrostatic levitation technique. We were able to determine the distribution of the electronic momentum density for the first time. From a detailed analysis of the distribution, we discovered an interesting fact: the distribution of the electronic momentum density of liquid silicon just above its melting point differs greatly from the predicted distribution based on simple atomic-structure assumptions. The distribution observed was very close to that of crystalline silicon. This suggests the high possibility that liquid-silicon atoms are not perfectly randomly arranged like those in ordinary simple liquids, but that liquid silicon has local structures very similar to crystalline silicon (Fig. 2). 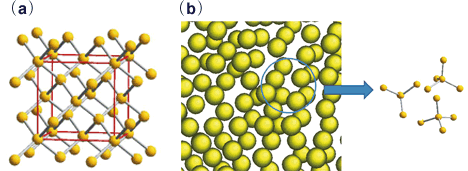
Conclusion The electrostatic levitation dissolution technique is an experimental technology that was first completed for a major aim, i.e., the ISS. We performed the experiment using SPring-8 as a spin-off. As a result, we were able to obtain an answer to the long-discussed question of the electronic structure of liquid silicon. Although we lack the space here to introduce them, we also obtained interesting results regarding other ultra-high-temperature liquids including liquid boron (melting point 2,180 deg. C). The electrostatic levitation and dissolution system is a very useful experimental tool that enables previously impossible research on ultra-high-temperature liquids. The electrostatic levitation and dissolution system was developed for the purpose of conducting experiments on the ISS. As research using the system on the ground has advanced, it has become clear which experiments are possible on the ground and which are possible only in space. For a single-ingredient liquid like silicon, we can measure on the earth with considerable accuracy, although minute influence of heat convection is unavoidable. In addition, since a large experimental facility like SPring-8 is unavailable on the ISS, ground experiments are important to complement space experiments. Materials such as oxide, fluoride and nitride are now highlighted as heat-resistant, functional or energy-saving materials. When heating these materials within the vacuum atmosphere required by the electrostatic levitation and dissolution system, however, gases are emitted so we cannot melt them. To melt them, we need the ISS that allows an experiment in a gas atmosphere. Therefore, we hope strongly that we will be able to install the system in the ISS and conduct melting experiments in a gas atmosphere. Until now, no such experiment has been performed in a gas environment. We may discover interesting phenomena beyond our imagination. There are many world-class facilities in Japan other than SPring-8. We intend to advance our research toward material development breakthroughs making full use of such facilities. The above research was conducted with the collaboration of researchers of JAXA, University of Tokyo, and Japan Synchrotron Radiation Research Institute (JASRI). I would like to express my sincere appreciation to both. Junpei T. OKADA |
| | 1 | 2 | 3 | |




