TOP > Report & Column > The Forefront of Space Science > 2004 > New Developments in Space Science>
![]()

Crystal growth-mechanism research in living-organism polymers In this research, we studied microscopic crystal-growth mechanisms at the atomic and molecular level where many scientific issues remain to be solved, in addition to macroscopic modeling in semiconductor crystal growth. Proteins were used as the material because they consist of a giant molecule that is relatively easy to observe and also is a familiar and important material for Mankind. For instance, living organisms produce about 100,000 kinds of proteins. An investigation of the molecular structure and function of proteins enables us to develop new drugs, foods, diagnoses, and medical treatments. Crystal-structure analysis by X-ray is usually used for the structural study of protein molecules. However, accurate structure determination requires high-quality protein crystals. It is thought that convection control is the most effective for high-quality crystal growth. Space experiments on more than 10,000 samples have been conducted including experiments by overseas space agencies but the success ratio for producing high-quality crystals is still less than 50 %. Thus, a study of the mechanism is necessary to improve quality. As stated above, since the molecular weight of a protein is about 10 thousands to hundreds of thousands, observation at molecular level is easy. Furthermore, depending on the sample, it is possible to observe a single molecule. We believe that by using the Atomic Force Microscope (AFM), it is possible to observe microscopic behavior such as the uptake of protein molecules into the growing boundary face. At present, we are using AFM to conduct crystal-surface observation and measurement of step progress speed, to name but a few. The measurement of boundary-face density, observation of crystal defects, and quality assessment by X-ray, are also being performed in parallel to systematically unveil the mechanism. Fig 3 shows one of the results obtained so far. The rough crystal surface is caused by ingestion of impurities.  Fig.3 Surface of a crystal when an impurity is mixed into it.
Scientific quest using non-contact flotation technique Under microgravity, we can hold material in a floating condition without a container. Using this advantage, we can easily create a non-coagulation state (supercooled state) even though the material is cooled from its melting state to below its coagulating temperature (melting point). When material is coagulated in a supercooled state, so-called metastable phases can occur, resulting in new, amorphous materials. Generally, the defining factor for material property is atomic arrangement and structure. Material with a metastable atomic arrangement and structure can exhibit new properties. The development of new metastable materials is expected for applications in laser and magnetic materials. The containerless technique also allows us to measure property values for highly reactive materials over wide temperature range, well above the melting temperature et under supercooled conditions. I will introduce below the research and development status of the electrostatic levitation furnace, a device to control the position of material in space using electrostatic field, and some of the scientific results obtained with the instrument. This is expected to be a next-generation experimental tool on the ISS. High-accuracy thermophysical property measurements research The electrostatic levitation furnace can position millimeter-size samples even on the ground. Research and development of a ground-based electrostatic levitation furnace is currently being performed. At present, it is possible to measure viscosity, surface tension, density, and isobaric specific heat of liquid-state materials, including their temperature dependency, up to about 3,000 deg C. Fig 4 shows the world's first measurement results of the viscosity of a metal (niobium, melting point of 2,500 deg C). Since this molten metal has a high reactivity, property measurement was difficult. Furthermore, we succeeded in measuring properties at a temperature 500 deg C lower than the melting point. Such a supercooled state cannot be attained by the traditional method involving a crucible. On the ground, errors in measuring property values occur because the sample size is limited and the sample shape is deformed from a perfect sphere due to gravity. Thus, it is expected that improved property measurement could be performed in microgravity. 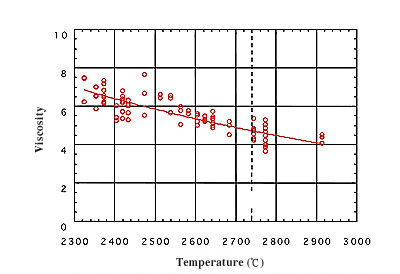 Fig. 4 Measurement result of viscosity of niobium (melting point: 2,470 deg C).
|
|||




