TOP > Report & Column > The Forefront of Space Science > 2005 > Gravity-Adaptive Systems of Living Things on Earth
![]()

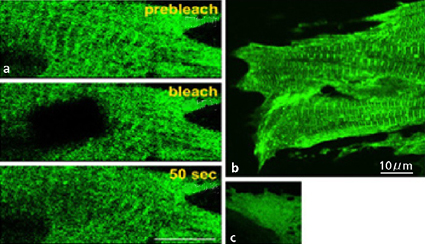
The molecular chaperone takes care of the protein system While maintaining structure to generate functions, life systems function by circulating two aspects with different directionality in various tires. The come-and-go between both poles becomes the dynamic-maintenance factor, or stress factor, to the cells and facilitates adaptation. This dynamic is the synthesis and decomposition of the protein itself: dynamic formation retention by copolymerization and depolymerization of cytoskeleton protein; consumption and generation of energy; form retention and tension exertion; contraction and relaxation (extension); exercise and structure maintenance; and stabilization and destabilization. Thus, cells regularly use functions of the regeneration system by the central dogma within one-generation lifespan. And cells entrust the maintenance of normal protein function in the regeneration system itself to the molecular chaperone (stress protein), which takes care of the protein’s lifespan. If protein denatures from this circulating system, it shifts to various clinical states (e.g., Alzheimer’s and prion disease). It is proved that 30% of the cell’s neo-genesis protein is abnormal, as it decomposes soon after it is synthesized. This evidence shows that the cell system must be continually, dynamically in motion. There are many stress proteins. In heart-muscle and slow-muscle cells, which are models for long life-adaptation strategy, there is a high expression rate of low-molecular-weight protein (sHSPs), among others, operating in the key part of the function/structure linkage system of energy dependency. One sHSPs, αB-crystallin, decreases specifically in a rat hind-leg suspension model, a zero-gravity muscle-atrophy model. In a space environment, if production of the stress protein (the cell’s adaptive-acquisition molecule obtained under gravity) becomes low, the continued stimulation of the adaptation system in living things, including humankind, will become a major problem. A mouse knocked out by αB-crystallin does not die immediately, but measurement shows extremely low levels in its motor activity. When expression of thermal-shock factor (the stress protein’s transcriptional control element) is forced on C elegans, its life almost doubles. Thus, it has been reported that the stress protein, as with IGF-1, is a longevity element. In this model, the stress protein that works to constrain the aggregation of denaturation proteins is another sHSPs, like αB-crystallin. Dynamic response to the cell’s interior and exterior and the molecular chaperone αB-crystallin and HSP47 On the ground, gravity is a constitutive factor. I considered a cell’s continuous dynamic response system by using the molecular chaperone (Fig. 2). I examined: the molecular mechanism of αB-crystallin on cytoskeleton protein that generates the tension exertion and contraction mechanism inside the cell; the gravity load of the chaperone HSP47 on the main protein collagen in the extra-cellular matrix, that is essential for fulcrum formation outside the cell; and response to release (rat model). HSP47 is a sole molecular chaperone, the expression of which is controlled by HSF inside endoplasmic reticulum. It was proved that HSP47 is essential for three-spiral polymerization of collagen protein, and for adding and processing secreted hydroxyl to the exterior of the cell.
With regard to the amount of HSP47 expressed, observations indicate that protein and mRNA respond to over-gravity by hind-leg suspension and centrifugal force much earlier than their ground substance, mRNA of collagen. Collagen is believed to be synthesized with fibroblast. Muscle cells have a basement membrane structure, however, and it was proved that it is synthesized within muscle cells as a result of studies using cell-culture systems.
|
|||||