TOP > Report & Column > The Forefront of Space Science > 2005 > Gravity-Adaptive Systems of Living Things on Earth
![]()

In the life-science field of space-science research, one of the important themes is a building theory of life systems produced by the earth itself and their evolution/adaptation mechanisms that have been at work for over 3.8 billion years. The life system’s strategy in pursuit of maintenance against stress can suggest hints about the existence of human beings, who have developed scientific and technological methods to cope with stress. Among the items I will introduce here are: structural adaptation systems and cytoskeletal proteins, which have been under performance analysis as key molecules in the gravitational adaptation of living things with form – especially mammals including human beings – that seem to be constructed as a system principle; and molecular chaperones (proteins that take care of proteins) essential for temporal adaptation of the cytoskeletal protein. These teach us that adaptation is acquired by bringing out skillfully the body’s stress response system. The core stress factors causing adaptation are gravitational and mechanical stimuli. Mechanical and dynamic systems giving form to living things Just as foundations and pillars are necessary to build a house on the ground, formed cells have a dynamic foundation, an extra-cellular matrix consisting of proteins, and a cytoskeleton, the raw materials that form them. The strength (tension) exerted by the cytoskeleton differs with the cells. When the cytoskeleton is destroyed or supplied with material that prevents its dynamics, the cell dies. Cell death also occurs when cells are peeled off their foundations, the extra-cellular matrix. Bioinformation is written in the genome, but it does not build forms. To read a genome, the field requires mechanical strength. To endure that strength, living things build a dynamically balanced structure with proteins or sugar outside and inside the cell.
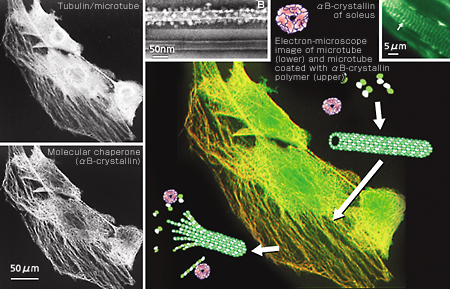
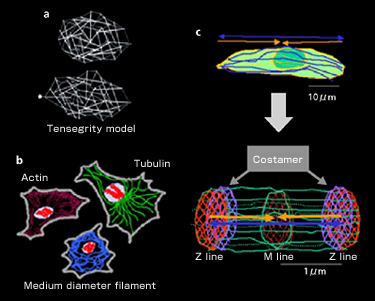
The cytoskeleton is a fiber structure to generate expansive or contractive elasticity and, at the same time, to resist that elasticity. Actin filament in cultured cells produces stress fiber that has a contractive structure similar to the sarcomere structure of muscles. D. Ingber of Harvard University named this dynamic structure the “tensegrity model” (Fig. 1-a), since it controls by tension as in a tent or Fukuoka Dome, and applied it to cells. All cells have three kinds of protein fiber, or cytoskeleton, in common: actin, tubulin and medium-diameter filament (Fig. 1-b). Contrary to the names “tensegrity” or “skeleton,” these are dynamically reproduced. In particular, the hollow nano-fibers and microtubes produced by tubulin reveal so-called dynamic instability. As the name indicates, the nano-fibers and microtubes combine and co-polymerize GTP, extending the microtubes. Once GTP is decomposed to GDP, however, it becomes unstable and starts depolymerizing and contracting. This results in an increase of non-polymerizing free forms, causing the extension to occur again. The maintenance of these chemically balanced relations forms the background to the dynamic instability. Further, once combined with increased free forms, tubulin’s mRNA is destabilized and destroyed. It becomes unable to produce tubulin. This suggests that the cells’ dynamic state is adjusted by proteins working at site in co-polymerizing/de-polymerizing states, rather than by self-control at genetic level. It is known that the microtube divides chromosomes into two during cell division. It has been proved that the microtube is essential in skeletal muscle cells for maintaining polarity at the time of fusion from myoblast to muscle fiber. Recently it was also proved that it maintains the dynamics of the cytoskeleton itself by interacting with the actin filament. It is thought that the actin filament provides muscle cells with contraction force, in particular, when interacting with myosin while the microtube counteracts with extension force (Fig. 1-c). Within this dynamic environment of mutual different directionality, cells do not move but sustain a tension-generating environment. In the sarcomere, it is highly structured (Fig. 1-c, bottom).
|
||||||||